Hardly a day goes by when a new digital health product doesn’t hit the market. Despite so many options, adoption of digital products into healthcare remains low. A lack of high-quality evidence supporting these products is one reason for this.
Digital therapeutics (DTx) are products within digital health that aim to impact disease outcomes. The Digital Therapeutics Alliance defines digital therapeutics as “evidence-based therapeutic interventions driven by high-quality software programs to prevent, manage, or treat a medical disorder or disease”. Evidence, or the proof, for a product involves information showing what kind of an effect it has, who it influences, and how long this effect is expected to last. DTx product claims of preventing, managing, and treating disease means their supporting evidence is compared to traditional medical innovations like pharmaceutical products. Unlike pharmaceutical products, where evidence demonstrates whether a medication is working, DTx evidence needs to demonstrate whether the digital product is not just working but is working for the specific people and in the specific life context for which it claims to have an effect. A medication’s effect does not depend on whether you take it in the office or at home. The effect of the medication reminder you receive on your phone or smartwatch, does.
Evidence quality is determined not by the effect demonstrated, but by the rigor of the process that delivered the effect. DTx product evidence involves information on both the science of the disease it aims to have an effect on, and the design of how this effect is achieved in the product experience. This means rigor is essential to demonstrate in both the scientific and design evidence of a DTx product. The first is a familiar type of evidence for healthcare experts to evaluate, the latter is not. Rigor in a scientific research process means applying a systematic and transparent methodology with complete reporting. Rigor in a product design process means the same, but of the two, is the more likely to be overlooked or compromised on. When rigor in the design process of DTx products is not held to the same high standard as its scientific research process, the product cannot demonstrate convincing and high-quality evidence for half of what makes up its effect: who it influences and the life context in which it works. For DTx products to be recognized as reliably preventing, managing, or treating disease, design process rigor must be equally attended to as scientific research rigor.
A rigorous design process creates the highest likelihood of the DTx product being “usable”. Usability means that a person knows how to use the product to achieve their goals with ease. With pharmaceutical products, usability factors into your medication prescription and the ease with which you can follow it. Do you know which medication you have to take, when you must take it, and how? Do you take it with food or without? Do you take it as a tablet or an injection? Having clear information answering these questions makes a medication “usable”. With DTx products, the concept is the same, but achieving it is far more difficult. For example, a blood pressure management smartphone app is usable if it fits into the flow of your day in terms of when you interact with it, clearly and understandably communicates your blood pressure readings and their interpretations, helps you remember to take your blood pressure medication throughout the day, gives you recommendations on how you can further improve your heart health with diet and exercise, and it does this over months or years, considerate of the health and life changes you go through. Usability is so essential to DTx product effectiveness, that it is regulated at international and local levels. Evidence of usability makes up a key part of the information supporting DTx product adoption and relies on a rigorous design process that follows regulatory guidance.
Establishing a rigorous DTx product design process increases the likelihood of achieving a usable product, but for the product to be adopted, usability has to be demonstrated as a part of the supporting evidence. Usability evidence can be demonstrated with evaluation research where the degree of usability is determined using methods that either center the user (inquiry) or expert reviews (inspection). Ten usability characteristics are globally recognized: accessibility, aesthetics, appropriateness/usefulness, effectiveness, efficiency, user error protection, learnability, memorability, satisfaction, and operability. Demonstrating any of these characteristics is only possible with a rigorous design process as this makes DTx product decisions measurable. Measurable usability in DTx products creates the evidence necessary to demonstrate that the product can prevent, manage and treat disease by showing it aligns with user health goals, users understand their health information, and they know what to do to with it.
Takeaways from designing two DTx candidates
The Newel Health product design team has spent the last two years designing two digital health solutions (DTx candidates), one for hypertension and the other for Parkinson’s Disease management. Here are our key takeaways of establishing this rigorous design process.
- Establish process reporting and evaluation documentation as early as possible to create opportunities for measuring design impact on usability characteristics.
- Initiate internal stakeholder alignment on product versions, key performance indicators, and research or testing strategies to create a common understanding of where the team, the company, and the product is at any given time. This will help clarify product versions, guide research and evidence generation decisions, and keep a rigorous design process central to the development of the product.
- Elevate user-centered outcomes in each version of the product and related evidence discussions, providing training to internal stakeholders where helpful, and adding user-centered outcomes to all research endeavors to the extent possible. This will create a mindset shift for internal stakeholders and will add to the reportability of the design research process.
- Advocate for consistent user research throughout each version of the product to mitigate the risk associated with assumptions and biases. This will help to find a balance between the business objectives and the users’ context.
- Advocate for documenting, tracking, and reporting of the design process, design research, and product decision-making to ensure adherence to regulatory guidelines, increasing design rigor, and elevating design impact evidence quality.
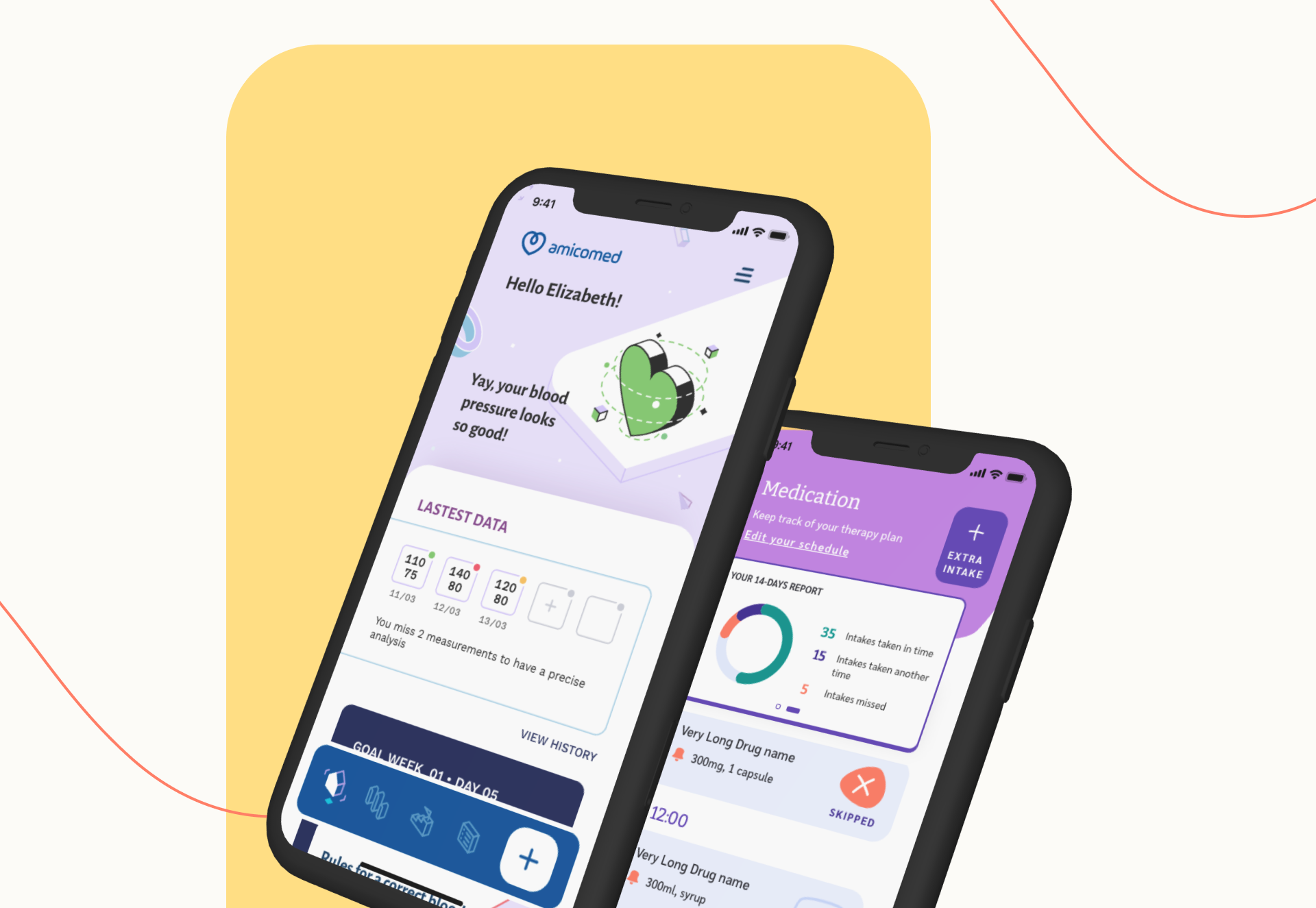
How is your product design team generating DTx product evidence capable of galvanizing adoption? Reach out to continue the conversation! Check out our digital health solutions (DTx candidates), Amicomed, designed for hypertension and Soturi, designed for Parkinson’s Disease.
Be sure to also look out for our follow-up articles in this collection of posts on our design process, including accessible UI and design requirements for specific user populations.
References
- Digital Therapeutics Alliance. https://dtxalliance.org (2020)
- Trust through evidence. Evidence generation for AI solutions in healthcare. https://www.provahealth.com/ph-insights (2023)
- Jessica L. Johnson, Donna Adkins, Sheila Chauvin, A Review of the Quality Indicators of Rigor in Qualitative Research, American Journal of Pharmaceutical Education, Volume 84, Issue 1, 2020, 7120, ISSN 0002-9459, https://doi.org/10.5688/ajpe7120. (https://www.sciencedirect.com/science/article/pii/S0002945923016066)
- Gondi, S., Powers, B.W. & Shrank, W.H. Evidence and Efficacy in the Era of Digital Care. J GEN INTERN MED 37, 2559–2561 (2022). https://doi.org/10.1007/s11606-022-07445-0
- DTx Product Quality & Access Worksheet. Digital Therapeutics Alliance (2020). https://dtxalliance.org/wp-content/uploads/2021/01/DTx_Quality_Access_Utilization_Worksheet.pdf
- Colin A Espie, Jenna R Carl, Richard Stott, Alasdair L Henry, Christopher B Miller, Digital medicine needs to work, The Lancet, Volume 392, Issue 10165, 2018, Page 2694, ISSN 0140-6736, https://doi.org/10.1016/S0140-6736(18)32519-4. (https://www.sciencedirect.com/science/article/pii/S0140673618325194)
- Patel, N.A., Butte, A.J. Characteristics and challenges of the clinical pipeline of digital therapeutics. npj Digit. Med. 3, 159 (2020). https://doi.org/10.1038/s41746-020-00370-8
- Wang, C., Lee, C. & Shin, H. Digital therapeutics from bench to bedside. npj Digit. Med. 6, 38 (2023). https://doi.org/10.1038/s41746-023-00777-z
- Christina Synowiec, Erin Fletcher, Luke Heinkel, Taylor Salisbury. Getting rigor right: a framework for methodological choice in adaptive monitoring and evaluation. Global Health: Science and Practice Dec 2023, 11 (Supplement 2) e2200243; DOI: 10.9745/GHSP-D-22-00243
- Maqbool, B., & Herold, S. (2024). Potential effectiveness and efficiency issues in usability evaluation within digital health: A systematic literature review. Journal of Systems and Software, 111881.(https://www.sciencedirect.com/science/article/pii/S0164121223002765)
- International Organization for Standardization. (2018). Ergonomics of Human System Interaction — Part 11: Usability: Definitions and Concepts.
- ISO 14971- International Organization for Standardization. (2019) Medical devices – Application of risk management to medical devices
- IEC 62366-1- International Electrotechnical Commission. Medical devices — Part 1: Application of usability engineering to medical devices.